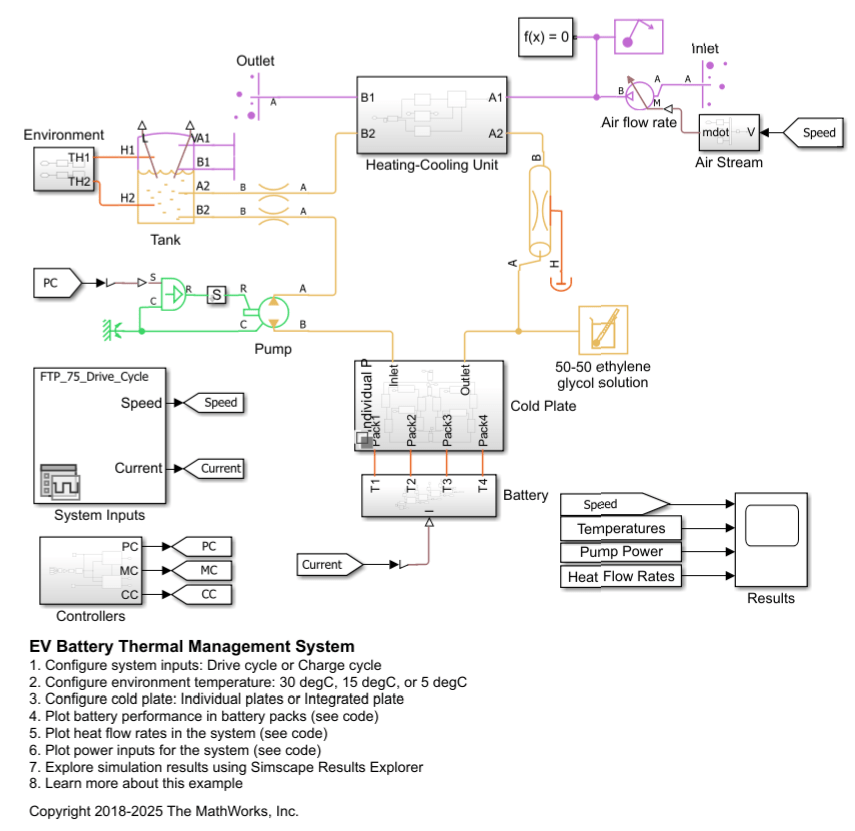
Thermal Liquid Properties (TL)
Preset fluid properties for the simulation of a thermal liquid network
Libraries:
Simscape /
Fluids /
Thermal Liquid /
Utilities
Description
The Thermal Liquid Properties (TL) block sets the predefined fluid properties of a thermal liquid network. The available fluids include pure water, aqueous mixtures, diesel, aviation fuel jet A, and SAE 5W-30. You can use this block as an alternative to the Thermal Liquid Settings (TL) block. If your network does not have a liquid properties block, the liquid defaults apply.
You can set values for density, the bulk modulus and thermal expansion coefficient, the specific internal energy and specific heat, the kinematic viscosity, and the thermal conductivity. The values you can enter for each block parameter depend on the liquid that you model. If you enter a value outside the valid range for a liquid property, the model generates an error during simulation.
Data Visualization
To plot the data, open the block dialog box and, under Plots, click the Plot button next to Thermal liquid properties. When you select a fluid property from the drop-down list, the plot updates. If you change the block parameter values, you can regenerate the plot by clicking Reload Data.
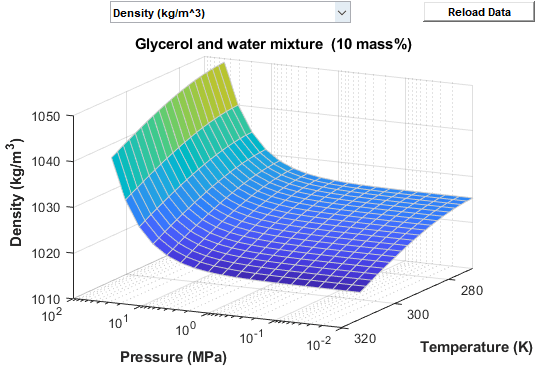
Regions of Valid Fluid Properties
The thermodynamic correlations that calculate fluid properties within the Thermal Liquid Properties (TL) block define the region of valid fluid properties. When you enter a value outside the valid ranges, the model generates an error during simulation. You cannot change the valid fluid properties region for each fluid. To visualize these regions, open the block dialog box and, under Plots, click the Plot button next to Thermal liquid properties.
The fluid properties of water are valid at temperatures between the triple point, 273.160 K, and the critical point, 647.096 K. They are valid at pressures between the triple point, 611.657 Pa, and the saturation pressure up to the critical point of 22.065 MPa. Water properties below the saturation point for a given temperature are not valid.
The fluid properties of seawater are valid at temperatures between 273.15 K and 393.15 K. They are valid at pressures above the saturation pressure and below 12 MPa. Pressures below the saturation point for the given temperature and concentration level are not valid. Mixture concentrations can range from 0 to 0.12 on a mass-fraction basis.
The fluid properties of an aqueous ethylene glycol mixture are valid between temperatures that depend on the mixture concentration and the freezing point of the liquid. You set the Minimum valid pressure and Maximum valid pressure in the block dialog box. Mixture concentrations can range from 0 to 0.6 on a mass-fraction basis, or from 0 to 1 on a volume fraction basis.
The fluid properties of an aqueous propylene glycol mixture are valid between temperatures that depend on the mixture concentration and the freezing point of the liquid. You set the values of Minimum valid pressure and Maximum valid pressure in the block dialog box. Mixture concentrations can range from 0 to 0.6 on a mass-fraction basis, or from 0 to 1 on a volume fraction basis.
The fluid properties of an aqueous glycerol mixture are valid between temperatures that depend on the mixture concentration and freezing point of the liquid. You set the Minimum valid pressure and Maximum valid pressure in the block dialog box. Mixture concentrations can range from 0 to 0.6 on a mass-fraction basis. The block smooths and extrapolates data to 100 degC depending on the type of data:
Extrapolation Method for Glycerol and Water Mixture
| Fluid Property | Extrapolation Method |
|---|---|
| Density | Quadratic |
| Thermal expansion coefficient | Linear |
| Specific heat | Linear |
| Specific internal energy | Linear |
| Kinematic viscosity | Exponential |
| Dynamic viscosity | Exponential |
| Prandtl number | Exponential |
The fluid properties of Jet-A fuel are valid at temperatures between 222.22 K and 645.1 K. They are valid at pressures between the saturation point and 2.51 MPa. Jet-A fuel properties below the saturation point for a given temperature are not valid.
The fluid properties of diesel fuel are valid at temperatures between 238.20 K and 690.97 K. They are valid at pressures between the saturation point and 2.29 MPa. Diesel fuel properties below the saturation point for a given temperature are not valid.
The fluid properties of SAE 5W-30 are valid at temperatures between 235.15 K and 473.15 K. They are valid at pressures between 0.01 MPa and 100 MPa. The block smooths and extrapolates data to cover all temperatures outside 303.00 K and 348.00 K and pressures outside 7 MPa and 82 MPa depending on the type of data:
Extrapolation Method for SAE 5W-30
| Fluid Property | Extrapolation Method |
|---|---|
| Density | Quadratic |
| Bulk Modulus | Linear |
| Thermal expansion coefficient | Linear |
| Specific heat | Linear |
| Thermal conductivity | Linear |
| Kinematic viscosity | Analytical |
| Dynamic viscosity |
|
| Prandtl number | Analytical |
Calculations of Fluid Properties
The block calculates the density and expansion coefficients using block parameters when you set Thermal liquid fluid list to one of these values:
Ethylene glycol and water mixturePropylene glycol and water mixtureGlycerol and water mixture
The block calculates the temperature and pressure dependent fluid density as
where:
T is the network temperature.
p is the network pressure.
ρ is the fluid density.
pR is the reference pressure associated with the fluid property tables.
ß is the isothermal bulk modulus.
The block calculates the change in fluid density with respect to temperature as
The thermal expansion coefficient is calculated as:
Model Entrained Air
If the value of the Volumetric fraction of entrained air in mixture at atmospheric conditions is nonzero, the thermal liquid network connected to the Thermal Liquid Properties (TL) block contains a small amount of nondissolved air. To model air dissolution, select the Model air dissolution check box. For more information, see Parameterizing Entrained Air in a Thermal Liquid.
Examples
Ports
Conserving
Parameters
References
[1] Massachusetts Institute of Technology (MIT), Thermophysical properties of seawater database. http://web.mit.edu/seawater.
[2] Nayar, Kishor T., et al. "Thermophysical Properties of Seawater: A Review and New Correlations That Include Pressure Dependence." Desalination, vol. 390, July 2016, pp. 1-24. DOI.org (Crossref), https://doi.org/10.1016.j.desal.2016.02.024
[3] Sharqawy, Mostafa H., et al. "Thermophysical Properties of Seawater: A Review of Existing Correlations and Data." Desalination and WaterTreatment, vol. 16, no. 1-3, Apr. 2010, pp. 354-80. DOI.org (Crossref), https://doi.org/10.5004/dwt.2010.1079